An In-Depth Exploration of the Electrochemical Oxygen Reduction Reaction (ORR) Phenomenon on Carbon-Based Catalysts in Alkaline and Acidic Mediums
Jul 19, 2022·,,·
0 min read
Niladri Talukder
Lead Author
,Yudong Wang
Bharath Babu Nunna
Eon Soo Lee
Abstract
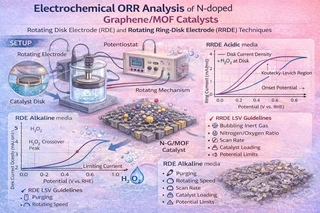
Detailed studies of the electrochemical oxygen reduction reaction (ORR) on catalyst materials are crucial to improving the performance of different electrochemical energy conversion and storage systems (e.g., fuel cells and batteries), as well as numerous chemical synthesis processes. In the effort to reduce the loading of expensive platinum group metal (PGM)-based catalysts for ORR in electrochemical systems, many carbon-based catalysts have shown promising results and numerous investigations are in progress. Most studies report ORR performance as current density curves from rotating disk electrode (RDE) and rotating ring-disk electrode (RRDE) experiments using cyclic voltammograms (CV) or linear sweep voltammograms (LSV). However, interpretations are often ambiguous and can lead to misunderstanding of ORR pathways and inaccurate characterization. Here, we study ORR on nitrogen-doped graphene/metal-organic framework (N-G/MOF) catalysts via RDE/RRDE LSV in both alkaline and acidic media. Key components of the RDE/RRDE setup and practical considerations are delineated as guidelines for new researchers. We discuss LSV curve features and their relationship to ORR pathways, and demonstrate how hydrogen peroxide (H2O2) in alkaline electrolyte and acid concentration in acidic electrolyte influence ORR current density in line with possible pathways.
Type
Publication
Catalysts (MDPI)